- High Sensitivity & Precision
Detects low-concentration tear biomarkers with excellent reproducibility, supporting early and accurate diagnosis. - Foolproof Operation
Each test cassette features a unique barcode that is automatically scanned by the analyzer—eliminating user error and ensuring the correct protocol is followed for each test type. - Built-in Safety & Quality Control
The analyzer continuously monitors internal parameters and test validity, alerting the user to any issues. Test integrity is protected at every step. - Ease-of-Use
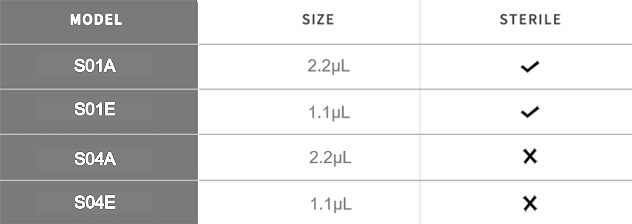
Minimal training required. Load the cassette, insert the sample (1–2 µL), and receive results within 10–15 minutes. Compact and portable for point-of-care flexibility. - Test-Specific Calibration
Barcode-linked protocols ensure each assay is run with precisely the right calibration curve and detection parameters—ensuring both standardization and accuracy. - Auto-Adaptation for New Tests
No software updates required. Every new test cassette carries its own encoded protocol, enabling seamless compatibility with the existing analyzer.
Why i-ImmunDx™ is Different:
Unlike immuno detection lateral, i-ImmunDx™ combines the clinical-grade rigor of a lab instrument with the speed and convenience of a point-of-care tool—bridging innovation and practicality
Two Test Modes:
Standard Test: Test cassette is immediately inserted into Analyzer after sample is added, so the user can multi-task during sample reaction time (10-15 minutes)
Quick Test: Test cassette is inserted after the reaction time is complete, allowing multiple tests to be performed in a single time frame(results in seconds).
– High-capacity storage
– Saves results automatically, allowing stored data to be easily aexported onto computers or recovered in the event of power failure.
– Built-in thermal printer for printing test results in real time
– Barcode scanner for easy identification of test cassettes and results
To purchase or request more information, send us an email at sales@seindabio.com or send us a message.